Nanostructured Materials: How Nano-Engineered Innovations are Transforming Solar Energy and Battery Technologies
- Green Fuel Journal

- Jan 2
- 40 min read
Nanostructured Materials: How Nano-Engineered Innovations are Transforming Solar Energy and Battery Technologies
Introduction
The global energy transition stands at a technological crossroads. Conventional silicon solar panels, despite decades of optimization, hover around 22-26% conversion efficiency in commercial applications—approaching their theoretical ceiling of approximately 29% dictated by the Shockley-Queisser limit.
Similarly, lithium-ion batteries face inherent constraints in energy density and charging speeds that impede their scalability for grid-scale storage and long-range electric mobility.
These limitations arrive at a moment when energy demands intensify: the International Energy Agency's Net Zero Emissions by 2050 Scenario requires global solar photovoltaic capacity to surge from approximately 1,185 GW in 2022 to over 5,000 GW by 2030—a nearly fourfold expansion within eight years. Simultaneously, battery energy storage must expand 30-fold to accommodate intermittent renewable generation.
Nanostructured materials—engineered substances with at least one dimension measuring between 1 and 100 nanometers—represent the scientific community's most promising response to these bottlenecks.
These materials exploit quantum mechanical phenomena, dramatically amplified surface areas, and tailored electronic properties to overcome fundamental limitations plaguing conventional energy technologies.
At the nanoscale, physics operates according to different rules: electrons exhibit quantum confinement effects, charge transport pathways become dramatically shorter, and surface-to-volume ratios increase exponentially.
A single gram of nanomaterial can possess a surface area exceeding 1,000 square meters, compared to less than 1 square meter for the same mass in bulk form.
The urgency driving nanomaterial innovation extends beyond incremental efficiency gains. Third-generation solar cells incorporating nanostructured materials have demonstrated laboratory efficiencies reaching 34.85% in perovskite-silicon tandem configurations, verified by the National Renewable Energy Laboratory (NREL) in 2025.
Quantum dot solar cells, leveraging size-tunable bandgaps, achieve 18.1% efficiency as of 2024 while targeting theoretical thermodynamic limits approaching 66% through hot carrier extraction. In energy storage, silicon nanowire electrodes deliver specific capacities exceeding 760 mAh/g—more than double graphite's theoretical 372 mAh/g—while enabling 5-10 times faster charge and discharge rates critical for electric vehicle adoption.
This comprehensive analysis examines how nanostructured materials are fundamentally transforming solar energy harvesting and electrochemical storage systems.
We evaluate the quantum mechanical principles enabling these breakthroughs, assess commercial readiness across different nanomaterial platforms, analyze synthesis methodologies from laboratory to manufacturing scale, and critically examine environmental implications of widespread deployment.
Our research team at GreenFuelJournal.com has synthesized findings from over 50 peer-reviewed studies, national laboratory technical reports, and industry roadmaps to deliver actionable intelligence for investors, policymakers, researchers, and sustainability officers navigating the intersection of nanotechnology and renewable energy.
What Are Nanostructured Materials and How Do They Work?
Nanostructured materials constitute a distinct class of engineered substances where material properties deviate dramatically from bulk counterparts due to dimensions constrained between 1 and 100 nanometers.
At this scale—roughly 1/1000th the width of a human hair—materials enter a regime where quantum mechanics governs behavior, classical physics predictions fail, and surface phenomena dominate over bulk characteristics.
The transition from bulk to nanoscale triggers three fundamental mechanisms that revolutionize energy applications: exponential surface area enhancement, quantum confinement of electronic states, and minimized charge transport distances.
The Physics Driving Nanoscale Advantage
Surface-to-Volume Ratio Enhancement represents the most immediately accessible benefit of nanoscale engineering. As particle dimensions decrease, the proportion of atoms residing at or near the surface increases exponentially relative to interior atoms.

A 10 nm nanoparticle possesses approximately 20% of its atoms at the surface, compared to less than 0.01% for a 1 mm particle of identical composition. This geometric transformation yields surface areas reaching 1,000 square meters per gram for optimized nanostructures—three orders of magnitude greater than bulk materials.
For photovoltaic applications, expanded surface area translates directly to increased photon absorption sites and enhanced light trapping through scattering mechanisms.
Research published in Frontiers in Energy Research (2025) demonstrates that nanomaterials can enhance incident solar radiation absorption by up to nine times, leading to 10% efficiency improvements in solar collectors compared to conventional designs.
In battery electrodes, amplified surface area provides dramatically more active sites for lithium-ion intercalation, enabling 5-10 times faster charging rates while maintaining capacity—critical for electric vehicle applications where rapid charging determines commercial viability.
Quantum Confinement Effects emerge when semiconductor dimensions shrink below the exciton Bohr radius, typically 2-10 nm for common photovoltaic materials. At these scales, electrons behave as waves rather than particles, and their allowed energy levels become discretized—similar to electrons in atoms rather than continuous bands in bulk semiconductors.
This quantum mechanical phenomenon enables unprecedented control over electronic and optical properties through size tuning alone.
Quantum dots—semiconductor nanocrystals typically 2-10 nm in diameter—exemplify this principle. A 3 nm cadmium selenide (CdSe) quantum dot exhibits a bandgap of approximately 2.4 eV, absorbing blue light and fluorescing blue. Increasing the diameter to 6 nm narrows the bandgap to roughly 1.9 eV, shifting absorption and emission to red wavelengths—all while maintaining identical chemical composition.
This size-tunable bandgap allows solar cell designers to optimize absorption across the solar spectrum by layering quantum dots of varying sizes, each harvesting specific wavelength ranges with minimal thermalization losses.
Recent research published in Nature Energy (2024) demonstrates that perovskite quantum dots synthesized through novel ligand exchange techniques achieve 15.3% power conversion efficiency while maintaining 83% of initial performance after 15 days of continuous operation. The quantum confinement in these sub-10 nm structures suppresses internal defects and enhances charge extraction compared to bulk perovskite films.
Enhanced Charge Transport through nanostructured pathways minimizes one of the most significant loss mechanisms in energy conversion devices: charge carrier recombination. In conventional polycrystalline solar cells, photogenerated electrons must navigate grain boundaries where 15-30% typically recombine before reaching collection electrodes.
Single-crystal nanowires provide direct, defect-free conduction paths spanning the active layer thickness.
Silicon nanowire solar cells fabricated at the National Institute for Materials Science demonstrate this advantage: the one-dimensional nanostructures enable shorter charge separation distances while maintaining sufficient optical path length through superior light trapping.
Research published in Frontiers in Nanotechnology (2024) shows that silicon nanowire cells achieve power conversion efficiencies approaching the Shockley-Queisser limit of 32% for single-junction devices while reducing material usage by over 60% through substrate thinning enabled by nanowire architectures.
Categories of Nanostructured Materials in Energy Applications
The nanomaterial landscape encompasses several distinct morphologies, each optimized for specific energy conversion or storage mechanisms:
Nanomaterial Type | Dimensional Structure | Typical Size Range | Primary Energy Application | Key Performance Advantage |
Nanoparticles (0D) | All dimensions <100nm | 2-50 nm | Plasmonic enhancement, dye sensitization | Tunable optical properties, high surface reactivity |
Quantum Dots (0D) | Spherical nanocrystals | 2-10 nm | Multi-junction solar cells, luminescent concentrators | Size-tunable bandgap (1.1-3.0 eV range) |
Nanowires (1D) | Length >>diameter | Diameter: 10-100 nm, Length: 1-100 μm | Direct charge transport, light trapping | Single-crystal pathways, minimal recombination |
Nanotubes (1D) | Hollow cylindrical | Diameter: 1-50 nm, Length: 1-100 μm | Electrode frameworks, transparent conductors | Exceptional conductivity, mechanical strength |
Nanosheets (2D) | Thickness <10nm | Thickness: 0.3-10 nm, Lateral: 100nm-10μm | Interface passivation, barrier layers | Controlled diffusion, selective permeability |
Nanocomposites (3D) | Multiple nanoscale components | Varies by architecture | Hybrid electrodes, protective coatings | Synergistic property combinations |
Nanoparticles and quantum dots dominate applications requiring optical property tuning. Gold and silver nanoparticles exhibit localized surface plasmon resonance—collective oscillations of conduction electrons at specific wavelengths determined by particle size and shape.
When integrated into solar cell absorber layers, plasmonic nanoparticles concentrate electromagnetic fields, enhancing absorption in adjacent photovoltaic materials.
Research from Abdullah Gül University (2024) demonstrates that spherical gold nanoshells deposited on organic solar cells boost light absorption by 36-66% depending on light polarization, providing angular coverage that captures photons across a wider incident angle range.
Nanowires and nanotubes excel in applications demanding efficient charge transport. Carbon nanotubes—cylindrical graphene sheets with diameters of 1-50 nm—possess electrical conductivities exceeding 10^6 S/m, rivaling copper while weighing a fraction as much.
When incorporated as transparent electrodes in solar cells or conductive additives in battery electrodes, carbon nanotubes reduce series resistance and improve rate capability.
Research published in Materials Advances (2024) shows that single-walled carbon nanotube (SWCNT) and zinc oxide nanostructure composites integrated into silicon thin-film solar cells achieve efficiency improvements through enhanced crystallinity and reduced surface defects.
The morphological diversity of nanostructured materials enables targeted optimization for distinct energy conversion steps: quantum dots tune light absorption, nanowires facilitate charge extraction, nanosheets passivate interfaces, and nanocomposites combine multiple functionalities.
Understanding these structure-property relationships guides rational nanomaterial design for next-generation renewable energy systems.
Nanostructured Materials in Solar Technologies
Solar photovoltaics face a fundamental challenge: converting the broad solar spectrum (spanning ultraviolet to infrared wavelengths) into electrical energy using materials with fixed bandgaps optimized for specific photon energies.
Single-junction silicon cells, despite decades of refinement, convert only approximately 26% of incident solar energy, losing the remainder to thermalization (high-energy photons), sub-bandgap transmission (low-energy photons), and various recombination mechanisms.
Nanostructured materials address these limitations through three complementary strategies: enhanced light absorption via optical engineering, bandgap tuning through quantum confinement, and minimized recombination through tailored charge transport pathways.
Enhancing Light Absorption Through Nanophotonics
Conventional planar solar cells reflect 30-35% of incident sunlight due to refractive index mismatches at the air-semiconductor interface.
Nanostructured surfaces transform this liability into an asset through sophisticated light management. Nanoscale surface texturing increases optical path length through scattering and waveguiding effects, effectively "trapping" photons within the absorber layer for multiple passes before escape.
Plasmonic nanoparticles leverage localized surface plasmon resonances to concentrate electromagnetic fields. When gold or silver nanoparticles of 30-100 nm diameter are embedded near solar cell absorbers, they act as nanoscale optical antennas. Incident light drives collective electron oscillations at resonance frequencies determined by nanoparticle size, shape, and composition.
These oscillations generate intense local electric fields that enhance absorption in adjacent photovoltaic materials by factors of 5-50 within nanometer-scale volumes.
Recent research published in Nanoscale Advances (2024) demonstrates that gold nanostructures combined with quantum dots in organic solar cells achieve 13% power conversion efficiency improvement—from 2.47% to 3.66%.
The quantum dots absorb ultraviolet photons and convert them to visible wavelengths through downconversion, while plasmonic gold nanoparticles enhance absorption in the visible spectrum. This dual-mechanism approach addresses spectral losses that limit conventional single-junction architectures.
Photonic crystals and metasurfaces represent more sophisticated nanophotonic approaches. These periodically structured materials with feature sizes comparable to light wavelengths manipulate photon propagation through engineered photonic band structures. Two-dimensional photonic crystals integrated onto thin-film solar cells create photonic bandgaps that inhibit reflection while recycling photons back into the absorber.
Research published in Frontiers in Applied Physics, Materials Science and Nanotechnology (2024) shows that dielectric metasurfaces composed of silicon or titanium dioxide nanostructures exhibit Mie resonances that enhance forward scattering while minimizing parasitic absorption losses, achieving broadband absorption enhancement across the visible spectrum.
Upconversion and downconversion nanoparticles extend spectral response beyond single-junction limitations. Rare-earth-doped nanocrystals can absorb two or more sub-bandgap photons and emit a single higher-energy photon (upconversion) or absorb high-energy photons and emit multiple lower-energy photons (downconversion).
While commercial implementation remains challenging due to low conversion efficiencies, laboratory demonstrations show promise for harvesting otherwise-wasted portions of the solar spectrum.
Key Nanomaterials Revolutionizing Solar Cell Architectures
Quantum Dot Solar Cells (QDSCs) exploit size-tunable bandgaps to optimize solar spectrum utilization. As of 2024, quantum dot solar cells achieve certified efficiencies of 18.1%, with theoretical thermodynamic limits approaching 66% through hot carrier extraction mechanisms.
The quantum dot solar cells market, valued at $910.49 million in 2024, is projected to reach $3,174.28 million by 2033 at a compound annual growth rate of 15.4%, reflecting investor confidence in the technology's commercial trajectory.
Lead sulfide (PbS) colloidal quantum dots have emerged as particularly promising for infrared harvesting. With bandgaps tunable from 0.7 to 1.5 eV through size control, PbS quantum dots access the near-infrared portion of the solar spectrum where approximately 50% of solar photons reside. Research from DGIST (2024) demonstrates that novel pulse-shaped light treatment of PbS quantum dot films enhances electrical conductivity while suppressing thermally induced surface traps, achieving rapid efficiency improvements without conventional high-temperature annealing that damages device interfaces.
Perovskite quantum dots combine bandgap tunability with exceptional defect tolerance. Research published in Nature Energy (2024) shows that organic cation-based perovskite quantum dots synthesized through advanced ligand exchange techniques achieve exceptional stability while suppressing internal defects.
These quantum dots deliver certified efficiencies exceeding 15% while maintaining performance under continuous illumination—addressing the stability challenges that have hindered commercialization of bulk perovskite films.
Silicon Nanowires reconcile two conflicting requirements: thick absorbers for complete light harvesting versus thin architectures for efficient charge collection. Vertically aligned silicon nanowire arrays with diameters of 50-200 nm and lengths of 1-10 μm provide optical path lengths exceeding 30 μm through light trapping while maintaining charge collection distances under 100 nm—dramatically reducing recombination losses.
Research published in Frontiers in Nanotechnology (2024) demonstrates that silicon nanowire single-junction solar cells fabricated on 280 μm substrates achieve power conversion efficiencies approaching the 32% Shockley-Queisser limit.
When substrate thickness is reduced to 100 μm through chemical etching, the nanowire architecture enables 64.5% weight reduction while maintaining efficiency—critical for applications in electric vehicles, aerospace, and building-integrated photovoltaics where weight constraints limit conventional silicon panels.
Perovskite Nanostructures have revolutionized third-generation photovoltaics. Metal halide perovskites with the general formula ABX₃ (where A = organic/inorganic cation, B = metal cation, X = halide anion) exhibit exceptional optoelectronic properties: high absorption coefficients exceeding 10⁵ cm⁻¹, long carrier diffusion lengths surpassing 1 μm, and bandgaps tunable from 1.2 to 3.0 eV through compositional engineering.
As of 2024, single-junction perovskite solar cells achieve certified efficiencies of 26.7% (University of Science and Technology of China), approaching the 27.8% record for silicon heterojunction cells while using 500 nm active layers versus 150-200 μm for silicon.
More significantly, perovskite-silicon tandem cells that stack perovskite top cells (bandgap ~1.68 eV) over silicon bottom cells (bandgap 1.12 eV) exceed the single-junction efficiency limit.
LONGi Green Energy achieved a certified 34.85% efficiency in 2025, verified by NREL—surpassing the 33.7% Shockley-Queisser limit for single junctions.
Recent breakthroughs address perovskite stability concerns. Research from the University of Surrey (2025) demonstrates that embedding aluminum oxide (Al₂O₃) nanoparticles within perovskite films traps mobile iodine ions that cause degradation. Perovskite solar cells with Al₂O₃ nanoparticle interfaces maintained high performance for over 1,530 hours under extreme heat and humidity—a tenfold improvement compared to 160 hours for unmodified devices. The nanoparticles create more uniform perovskite structures while forming protective two-dimensional perovskite layers that act as moisture barriers.
Similarly, Northwestern University researchers (2024) developed amidinium-based surface ligands that replace conventional ammonium coatings on perovskite nanocrystals. These more stable molecular structures are 10 times more resistant to decomposition under environmental stress. Solar cells with amidinium coatings achieved 26.3% efficiency while retaining 90% of initial performance after 1,100 hours of testing under harsh conditions—tripling the T90 lifetime (time to 90% efficiency retention) compared to conventional cells.
Case Studies: Laboratory Records Versus Commercial Readiness
The gap between laboratory efficiency records and commercial product performance reflects the multifaceted challenges of scaling nanomaterial photovoltaics.
Laboratory cells benefit from:
Small active areas (0.05-1.0 cm²) minimizing defect density
Controlled atmospheric conditions during fabrication
Optimized but expensive materials and processing
Commercial modules must deliver:
Large active areas (>1.6 m²) with uniform performance
Manufacturing throughput exceeding 5 GW annually
25-30 year operational lifetimes under field conditions
Installed costs below $0.50/W to compete with conventional silicon
Oxford PV represents the most advanced commercialization effort for perovskite tandems. The company's perovskite-silicon tandem panels achieve 24.5% module efficiency—exceeding the 22-24% typical for commercial monocrystalline silicon.
Oxford PV shipped modules to the United States in September 2024 for utility-scale installations and reports cells designed for 25-year lifespans based on over three years of outdoor testing.
Their Brandenburg, Germany factory targets gigawatt-scale production, with Chief Technology Officer Chris Case confirming cells lose only ~1% efficiency in their first year with minimal subsequent degradation—though detailed data remains proprietary.
Quantum dot solar cells face longer commercialization timelines. While companies like UbiQD develop luminescent solar concentrator windows using near-infrared quantum dots for building-integrated photovoltaics, standalone quantum dot cells remain confined to research laboratories. The challenges include:
Ligand engineering complexity: Quantum dot surfaces require molecular passivation to minimize trap states, but ligands must balance surface passivation with charge transport—often conflicting requirements
Ink formulation stability: Colloidal quantum dot inks must remain stable during storage and deposition while forming uniform films after drying
Inter-dot coupling: Quantum dots in films must be close enough for charge transport (typically <2 nm separation) while maintaining quantum confinement—requiring precise surface chemistry
Despite these hurdles, the $910 million global quantum dot solar cell market reflects sustained investment driven by theoretical efficiency advantages and compatibility with low-cost solution processing.
Nanostructured Materials in Battery and Energy Storage
Electrochemical energy storage confronts limitations fundamentally different from solar conversion: batteries must reversibly store and release charge through solid-state diffusion processes that scale poorly with device size.
Conventional lithium-ion batteries employing micrometer-scale electrode particles face trade-offs between energy density (requiring thick electrodes) and power density (favoring thin electrodes with short diffusion distances).
Nanostructured materials resolve this conflict by providing three-dimensional architectures that maintain high active material loading while offering nanoscale diffusion pathways and dramatically expanded electrode-electrolyte interface areas.
Three-Dimensional Electrode Architectures
The transition from planar to three-dimensional nanostructured electrodes represents one of the most impactful nanomaterial applications in energy storage. Conventional battery electrodes consist of 5-20 μm active material particles mixed with conductive carbon additives and polymer binders, cast as 50-150 μm thick films on metal foil current collectors.

During charge-discharge cycling, lithium ions must diffuse through these micrometer-scale particles—a process that limits charging rates to 0.5-2C (full charge in 30-120 minutes) without causing concentration gradients that degrade capacity.
Nanostructured electrodes replace particle-binder composites with three-dimensional frameworks where active materials are deposited as <50 nm coatings on high-surface-area conductive scaffolds. This architecture delivers several advantages:
Reduced Solid-State Diffusion Distances: Lithium-ion diffusion coefficients in electrode materials typically range from 10⁻¹⁰ to 10⁻¹⁴ cm²/s. Diffusion time scales with the square of distance, so reducing particle size from 10 μm to 50 nm decreases characteristic diffusion time by a factor of 40,000—enabling charge rates exceeding 10C (full charge in 6 minutes) while accessing full theoretical capacity.
Amplified Electrode-Electrolyte Interface: The total interface area where charge transfer reactions occur increases linearly with surface area. Nanostructured electrodes with surface areas of 50-200 m²/g provide 100-1000 times more interface than conventional micrometer-particle electrodes. This expanded reaction area reduces local current densities for a given total current, minimizing overpotentials and improving energy efficiency.
Accommodation of Volume Change: High-capacity electrode materials like silicon (theoretical 3,579 mAh/g—nearly 10 times graphite's 372 mAh/g) undergo >300% volume expansion during lithiation. In micrometer-scale particles, this expansion generates mechanical stresses exceeding 1 GPa that pulverize particles and sever electrical connections. Nanoscale architectures with sub-100 nm feature sizes remain below the critical fracture dimension, maintaining mechanical integrity through hundreds of cycles.
Research published in the International Journal of Low-Carbon Technologies (2024) synthesized findings across 1,000+ publications on nanotechnology for electric vehicle batteries. The systematic review identifies carbon nanotubes, graphene, and metal oxide nanostructures as the three most impactful material categories for improving energy density, charge time, and cycle life.
Key findings include:
Nanotubes and graphene improve energy storage capacity and ion flow through exceptional electrical conductivity (>10⁶ S/m) combined with high surface areas
Metal oxide nanostructures (manganese oxide, titanium dioxide) offer superior electrochemical stability and cycling performance compared to bulk counterparts
Nanoscale catalysts (platinum, palladium nanoparticles) enhance oxygen reduction/evolution reactions in fuel cells and metal-air batteries
Nanostructured solid electrolytes reduce lithium dendrite formation—a critical safety improvement over liquid electrolytes
Next-Generation Battery Architectures
Silicon Nanowire Anodes address the single greatest limitation preventing silicon from displacing graphite as the standard lithium-ion anode: mechanical degradation from volume expansion. Silicon nanowires with diameters of 50-200 nm synthesized through vapor-liquid-solid (VLS) or solution-liquid-solid growth methods maintain structural integrity through volume changes that would shatter micrometer-scale particles.

Research summarized in Journal of Power Sources demonstrates that silicon nanowire electrodes deliver reversible capacities exceeding 760 mAh/g for the first 10 cycles and maintain over 630 mAh/g after 50 cycles when cycled at 100 mA/g—representing 70-85% capacity retention. The tin-core/carbon-sheath coaxial nanocable structures described in recent literature combine 61% tin content with protective carbon coatings, achieving both high capacity and improved cycle stability.
Lithium Titanate (Li₄Ti₅O₁₂) Nanostructures sacrifice some energy density for exceptional power capability and safety.
While lithium titanate's theoretical capacity of 175 mAh/g significantly lags silicon or even graphite, its 1.55 V lithiation potential versus lithium metal eliminates lithium plating risks that cause internal short circuits in conventional cells charged too rapidly. This higher potential operates within the thermodynamic stability window of liquid electrolytes, avoiding electrolyte decomposition and harmful side reactions.
Critically, lithium titanate requires nanostructuring to achieve competitive performance due to poor intrinsic electronic and ionic conductivity. Research published in Nanotechnology confirms that nanocrystalline lithium titanate with grain sizes of 10-50 nm enables full theoretical capacity at high charge rates (>5C) through minimized solid-state diffusion distances.
The "zero-strain" characteristic of lithium titanate (negligible volume change during cycling) combined with nanoscale dimensions yields electrode materials capable of >10,000 cycles with minimal capacity fade—ideal for grid energy storage where cycle life outweighs energy density.
High-Entropy Oxides represent an emerging nanomaterial class for batteries. These complex oxides containing five or more metallic elements in near-equimolar ratios exploit configurational entropy to stabilize single-phase structures with synergistic properties.
Research published in Rare Metals (2024) highlights how high-entropy materials in battery electrodes provide:
Adjustable electrochemical properties through multi-element synergies
Enhanced structural stability from entropy stabilization preventing phase transformations
Improved cycling performance compared to traditional single or binary metal oxides
The concept extends beyond oxides to high-entropy alloys and multi-anionic compounds, offering new degrees of freedom in battery materials design. However, synthesis challenges and limited mechanistic understanding currently confine high-entropy nanomaterials primarily to research laboratories.
Supercapacitors: Bridging Batteries and Capacitors
Supercapacitors (electrochemical double-layer capacitors and pseudocapacitors) occupy the performance space between conventional capacitors (high power, low energy) and batteries (high energy, low power).
While energy densities of 5-15 Wh/kg lag lithium-ion batteries' 150-250 Wh/kg, supercapacitors deliver power densities exceeding 10 kW/kg and cycle lifetimes surpassing 1 million cycles—attributes valuable for regenerative braking, power quality management, and burst power applications.
Nanostructured carbon materials dominate supercapacitor electrodes. Activated carbon with pore sizes of 1-5 nm and surface areas of 1,000-3,000 m²/g stores charge through electrostatic adsorption of ions at the electrode-electrolyte interface.
Capacitance scales linearly with accessible surface area, making nanoporous carbons ideal for maximizing charge storage in minimal volume.
Graphene and carbon nanotubes offer superior alternatives to activated carbon. Graphene's theoretical surface area of 2,630 m²/g combined with exceptional electrical conductivity (10⁶ S/m) enables rapid charge-discharge. However, graphene sheets tend to restack during electrode fabrication, dramatically reducing accessible surface area. Strategies to prevent restacking include:
Chemical functionalization introducing hydroxyl, carboxyl, or other groups that impede sheet aggregation
Three-dimensional graphene aerogels where sheets crosslink into porous networks
Graphene-metal oxide nanocomposites where oxide nanoparticles physically separate graphene layers while contributing pseudocapacitive charge storage
Research published in Nanomaterials (2022) describes MnOₓ@graphite nanostructures used in symmetric pouch cell supercapacitors with solid-state polymer gel electrolytes. These devices achieve areal energy density of 7.87 mWh·cm⁻² and areal power density of 1,099.64 mW·cm⁻² with 2.2 V cell voltage—performance approaching lithium-ion batteries in energy density while maintaining supercapacitor-level power capability.
Pseudocapacitive materials store charge through fast, reversible faradaic reactions at electrode surfaces rather than bulk diffusion. Manganese dioxide (MnO₂) and ruthenium dioxide (RuO₂) are the most studied pseudocapacitive oxides, delivering 2-4 times higher capacitance than carbon alone. Recent work identified iron tungstate (Fe₂WO₆) as a novel pseudocapacitive material for negative electrodes in aqueous electrolytes.
When paired with MnO₂ positive electrodes, iron tungstate enables supercapacitors with enhanced energy density versus classical electrical double-layer capacitors based on carbon.
The convergence of battery and supercapacitor technologies through nanostructured materials yields "hybrid supercapacitors" combining battery-type anodes (offering high capacity) with supercapacitor-type cathodes (providing high power). Research published in Nanomaterials (2022) demonstrates ZnO nanorods grown directly on aluminum substrates functioning as high-power electrodes with excellent rate capability and >1,000 cycle stability—exemplifying binder-free, nanostructured architectures that maximize both energy and power density.
Synthesis, Fabrication, and Material Design
The translation of nanomaterial principles from laboratory demonstrations to commercial energy devices hinges critically on scalable, cost-effective synthesis methods.
While researchers often optimize nanomaterial properties using techniques incompatible with manufacturing—ultra-high vacuum systems, multi-step purifications, batch-to-batch inconsistencies—industrial deployment requires reproducible processes delivering kilograms to tons of uniform nanomaterials at costs below $100/kg for battery electrodes and $50/m² for solar cell components.
Synthesis methodologies bifurcate into top-down approaches that reduce bulk materials to nanoscale dimensions and bottom-up approaches that assemble nanomaterials from molecular or atomic precursors.
Top-Down Synthesis: Dimensional Reduction
Mechanical milling (ball milling) represents the simplest and most widely practiced top-down technique. High-energy collisions between grinding media (typically hardened steel or ceramic balls) and precursor powders progressively fracture particles down to nanoscale dimensions. While conceptually straightforward, mechanical milling suffers from:
Broad size distributions: Continuous fracture produces particles spanning 10 nm to 10 μm, necessitating post-synthesis classification
Surface contamination: Extended milling introduces wear debris from grinding media and atmospheric gases
Crystallographic damage: Mechanical stress generates defects, dislocations, and sometimes amorphization—degrading electronic properties
Despite these limitations, mechanical milling dominates for high-capacity anode materials like silicon where 70-80% cost reductions versus chemical synthesis methods justify accepting less-than-ideal particle morphology. Industry combines mechanical milling with subsequent annealing steps that repair crystallographic damage while maintaining nanoscale grain sizes.
Laser ablation uses pulsed laser beams to vaporize material from solid targets in controlled atmospheres. The vaporized species cool rapidly, condensing as nanoparticles with sizes determined by laser parameters (wavelength, pulse duration, fluence) and ambient gas pressure.
Laser ablation produces exceptionally pure nanoparticles free from chemical precursor residues but struggles with throughput—typical production rates of milligrams per hour confine the technique to research and specialty applications like fullerene synthesis for fundamental studies.
Lithography—patterning through selective material removal—dominates semiconductor device fabrication but plays limited roles in energy nanomaterials due to cost constraints.
Electron-beam lithography achieves sub-10 nm resolution but requires serial writing at <1 mm²/hour throughput, making it prohibitively expensive for square-meter solar panels or gram-scale battery materials.
Photolithography offers higher throughput but resolution limitations of 100-200 nm for deep-UV systems. Lithographic techniques find niche applications in photonic crystals for light management and precision nanoelectrode arrays for fundamental electrochemical studies.
Bottom-Up Synthesis: Molecular Assembly
Chemical Vapor Deposition (CVD) deposits solid materials from gaseous precursors through thermal, plasma, or photochemical activation. For nanomaterial synthesis, CVD produces:
Carbon nanotubes and graphene through catalytic decomposition of hydrocarbons over transition metal nanoparticles at 600-1000°C
Semiconductor nanowires via vapor-liquid-solid (VLS) growth where gaseous precursors dissolve in liquid metal catalyst droplets, precipitating as single-crystal nanowires
Conformal nanofilms for surface passivation and protective coatings
Atomic layer deposition (ALD)—a CVD variant using self-limiting surface reactions—enables precise thickness control at the sub-nanometer level. ALD's digital growth mode (one atomic layer per precursor cycle) allows coating complex three-dimensional nanostructures with uniform films—critical for solid-state electrolyte coatings on battery electrode nanomaterials.
However, ALD's low deposition rates (<1 nm/minute) and expensive organometallic precursors currently restrict applications to high-value components like protective coatings on silicon nanowire anodes.
Sol-gel synthesis converts liquid precursors (typically metal alkoxides dissolved in alcohols) into solid networks through controlled hydrolysis and condensation reactions. The sol-gel process is favored for metal oxide nanomaterials (TiO₂, ZnO, Fe₂O₃) used in photocatalysis, dye-sensitized solar cells, and battery electrodes due to:
Low processing temperatures (typically <500°C) reducing energy costs
Compositional control through stoichiometric precursor mixing
Scalability to batch sizes exceeding 100 liters
Recent reviews in Polymers (2024) and Journal of Chemical Reviews (2024) emphasize that sol-gel processes produce nanoparticles with superior homogeneity and purity compared to solid-state reactions.
The technique accommodates dopant incorporation at atomic scales, enabling precise bandgap engineering for photovoltaics. Disadvantages include relatively long processing times (gelation, drying, annealing spanning hours to days) and sensitivity to atmospheric moisture during synthesis.
Hydrothermal and solvothermal synthesis conduct reactions in sealed autoclaves at 100-300°C and autogenous pressures of 1-20 MPa. Elevated temperature and pressure increase precursor solubility and reaction kinetics, enabling crystalline nanomaterial formation without high-temperature annealing. Hydrothermal methods excel for:
Metal oxide nanostructures with controlled morphologies (nanorods, nanosheets, hierarchical architectures)
Chalcogenide quantum dots (CdS, CdSe, PbS) with narrow size distributions
Lithium-ion battery cathode materials (LiFePO₄, lithium transition metal oxides)
The technique's main limitation is batch processing: each autoclave run requires hours to days, and scaling requires multiple parallel reactors rather than continuous processing. Nonetheless, hydrothermal synthesis dominates commercial production of lithium iron phosphate cathodes for electric vehicles due to its ability to produce phase-pure, crystalline materials at <500°C—substantially lower than >700°C required for solid-state synthesis.
Colloidal synthesis (hot-injection method) delivers the highest-quality quantum dots with near-atomic size control. The technique rapidly injects cold precursors into hot coordinating solvents (200-350°C), inducing burst nucleation followed by controlled growth.
Monodisperse quantum dot populations with size standard deviations <5% enable precise bandgap tuning critical for optimized solar spectrum absorption.
Companies like Quantum Materials Corp employ continuous-flow adaptations of hot-injection synthesis targeting kilogram-scale production of perovskite quantum dots for photovoltaics—though cost competitiveness versus conventional thin-film deposition remains unproven.
Green and Biological Synthesis Approaches
Growing environmental consciousness drives interest in biosynthesis using biological entities as nanomaterial factories. Plants, fungi, bacteria, and algae naturally produce nanoparticles through metabolic reduction of metal ions coupled with biomolecule-mediated capping.
While primarily explored for biomedical applications (where biocompatibility and non-toxicity are paramount), green synthesis methods offer potential advantages for energy nanomaterials:
Ambient conditions eliminating energy-intensive heating
Non-toxic reagents from biological extracts versus harsh chemicals
Biocompatible surface chemistries potentially beneficial for solid-electrolyte interphase formation
However, practical limitations constrain biosynthesis adoption: slow reaction kinetics (hours to days), broad size distributions, and batch-to-batch variability from natural feedstock inconsistencies. As noted in Journal of Chemical Reviews (2024), green synthesis methods currently "work just as well as conventional synthesis techniques" for laboratory-scale exploratory research but lack the reproducibility and throughput demanded by commercial manufacturing. Continued development might overcome these hurdles, particularly for applications where sustainability attributes justify higher costs.
From Laboratory to Manufacturing: Scalability Challenges
The transition from milligram laboratory samples to kilogram commercial production confronts multiple technical and economic barriers:
Cost Structure: Many high-performance nanomaterials employ expensive precursors (organometallic compounds, high-purity metals, rare-earth elements).
While acceptable for $500/kWh specialty battery applications, solar modules targeting <$0.50/W installed costs demand materials priced well below $50/kg. Silicon nanoparticles produced through laser ablation might cost $500-1000/kg; mechanically milled silicon costs $20-30/kg—a stark difference that often determines commercial viability.
Throughput: Research synthesis methods optimized for material quality rarely scale efficiently. CVD reactors producing pristine nanowires at grams per hour cannot compete with silicon wafer production exceeding gigawatts annually.
Colloidal quantum dot synthesis delivering monodisperse populations in 100 mL batches must transition to continuous-flow reactors processing liters per hour while maintaining size control—an engineering challenge requiring sophisticated real-time monitoring and feedback control.
Quality Control: Nanomaterial properties depend sensitively on size, shape, crystallography, and surface chemistry—all potentially variable across production batches. Commercial energy devices require ±5% performance variation across modules to ensure system-level reliability. Achieving this consistency demands:
In-line characterization monitoring particle size distributions, crystalline phases, and chemical composition
Process parameter control maintaining precursor concentrations, temperatures, and reaction times within narrow windows
Post-synthesis classification separating desired nanomaterial populations from out-of-specification fractions
Companies like Oxford PV invest heavily in roll-to-roll processing that deposits perovskite layers onto silicon cells at speeds approaching meters per minute—industrial throughputs necessary for gigawatt-scale production.
The challenge lies in maintaining nanoscale control (grain sizes, interface quality) during high-speed processing, requiring innovations in real-time monitoring and adaptive process control.
Environmental, Safety, and Lifecycle Considerations
The environmental implications of widespread nanomaterial deployment span their entire lifecycle: raw material extraction, energy-intensive synthesis, application use-phase, and end-of-life disposal or recycling.
While nanostructured materials enable cleaner energy generation and storage, their production and potential environmental release raise questions demanding rigorous assessment.
The nanotechnology industry, valued at over $125 billion in 2024 and expanding rapidly, must reconcile innovation momentum with responsible development addressing worker safety, environmental persistence, and toxicological uncertainties.

Production Phase Environmental Footprint
Life-cycle analyses consistently identify the production phase as contributing the highest environmental and health impacts for engineered nanomaterials. Research published in Nature Reviews Earth & Environment (2024) synthesizes global nanotoxicology literature, concluding that "the highest potential environmental and health impacts of ENMs [engineered nanomaterials] used in commercial products are associated with production rather than incidental release during use."
Several factors drive production-phase impacts:
Energy Intensity: Many nanomaterial synthesis routes require elevated temperatures, high pressures, or ultra-high vacuum conditions. CVD processes for carbon nanotubes operate at 700-1000°C for hours, consuming 30-50 kWh per kg of product—substantially higher than 5-10 kWh/kg for commodity chemicals.
Sol-gel synthesis followed by calcination demands 400-800°C annealing, while colloidal quantum dot synthesis maintains reaction temperatures at 250-350°C for extended periods. Collectively, these energy inputs translate to carbon footprints that can offset environmental benefits if renewable electricity doesn't power production.
Chemical Precursors: High-purity precursors essential for many nanomaterial routes carry environmental burdens from their own production. Organometallic compounds used in CVD and ALD processes often involve energy-intensive synthesis, toxic intermediates, and hazardous waste streams. Metal nanoparticle production consumes metal salts refined through processes generating acidic wastewater and heavy metal residues.
Solvent Consumption: Colloidal synthesis and solution-based nanoparticle processing consume substantial volumes of organic solvents—toluene, hexane, chloroform, dimethylformamide—many classified as volatile organic compounds (VOCs) or hazardous air pollutants.
While solvent recycling can recover >90% in controlled industrial settings, laboratory-scale research often discards used solvents as hazardous waste.
Advancing toward sustainable nanomaterial production requires:
Renewable energy integration powering synthesis facilities with solar, wind, or hydroelectric electricity
Process intensification reducing reaction times, temperatures, and solvent volumes through catalytic pathways or continuous-flow reactors
Green chemistry principles substituting benign solvents (water, ethanol, supercritical CO₂) for hazardous organics where possible
Occupational Health and Worker Safety
Workers in nanomaterial production facilities face potential inhalation exposure to airborne nanoparticles during synthesis, handling, and packaging operations.
Particles <10 μm are respirable, reaching alveolar regions of lungs where removal mechanisms function poorly. Nanoparticles <100 nm exhibit enhanced biological reactivity due to their high surface area-to-mass ratios, potentially crossing biological barriers that retain larger particles.
Specific occupational health concerns include:
Pulmonary Effects: Certain nanomaterials—particularly high-aspect-ratio materials like carbon nanotubes and nanowires—exhibit fiber-like behaviors raising concerns analogous to asbestos. While carbon nanotubes are chemically inert, animal studies show that long, rigid nanotubes can induce pulmonary inflammation and fibrosis when inhaled at high doses.
The European Union's categorization of titanium dioxide (TiO₂) as a category 2 carcinogen due to inhalation hazards highlights regulatory attention to nanomaterial respiratory risks, though this classification applies specifically to inhalation of TiO₂ dust rather than nanoparticles embedded in solid products.
Dermal Exposure: Nanoparticle skin penetration remains debated, with most studies suggesting minimal transdermal uptake of inorganic nanoparticles through intact skin. However, mechanical abrasion, chemical exposure, or pre-existing dermatological conditions might facilitate penetration. Prudent workplace practices employ gloves and protective clothing regardless of definitive penetration evidence.
Engineering Controls: Properly designed facilities employ hierarchy of controls emphasizing elimination/substitution (using less hazardous nanomaterials), engineering controls (enclosures, ventilation, filtration), and personal protective equipment (respirators, gloves) as last resort.
HEPA filtration systems with 99.97% capture efficiency for 0.3 μm particles provide effective protection, though smaller nanoparticles exhibiting Brownian motion are also efficiently captured through diffusional mechanisms despite their size.
Research published in the Journal of Occupational and Environmental Hygiene (2013) documents that properly implemented engineering controls in U.S. nanotechnology workplaces effectively minimize worker exposure to levels well below occupational exposure limits. The challenge lies in ensuring smaller facilities—university laboratories, startup companies—maintain equivalent protection standards.
Environmental Fate and Ecotoxicity
Engineered nanomaterials inevitably enter environmental compartments through product lifecycle stages: manufacturing emissions, consumer product use, and end-of-life disposal.
Understanding their environmental behavior requires assessing:
Transformation and Aging: Released nanomaterials rarely maintain pristine properties in environmental matrices. Oxidation, sulfidation, dissolution, aggregation, and biomolecule corona formation alter surface chemistry, aggregation state, and biological interactions.
For instance, silver nanoparticles in antimicrobial textiles oxidize to silver sulfide (Ag₂S) in wastewater—a transformation dramatically reducing toxicity. Research in Environmental International (2015) demonstrates that nanomaterial aging and transformations through product lifecycles fundamentally alter exposure scenarios and risk profiles.
Bioaccumulation and Trophic Transfer: While conventional persistent organic pollutants accumulate through food webs following log Kow (octanol-water partition coefficient) predictions, nanomaterials exhibit more complex behaviors.
Most inorganic nanoparticles show limited bioaccumulation due to low lipid solubility and particle size constraints on cellular uptake. However, smaller nanoparticles (<20 nm) can cross biological membranes, and surface coatings modulate uptake.
The OECD Working Party on Manufactured Nanomaterials explores developing tiered approaches for bioaccumulation assessment specific to nanomaterials, recognizing that conventional models developed for molecular chemicals poorly predict nanoparticle behavior.
Ecotoxicity: Laboratory studies reveal nanomaterial toxicity varies enormously with composition, size, shape, and surface chemistry. Research published in Nature Reviews Earth & Environment (2024) notes that "typically, the detected or predicted ENM concentrations are 1 to 4 orders of magnitude lower than their respective predicted no-effect concentrations."
This substantial margin suggests environmental nanomaterial concentrations from current applications pose minimal ecological risk under typical exposure scenarios.
However, specific nanomaterials warrant continued monitoring:
Silver nanoparticles in consumer products show aquatic toxicity to algae and invertebrates at concentrations potentially achievable in contaminated surface waters
Titanium dioxide nanoparticles from sunscreens and paints demonstrate phototoxicity to aquatic organisms under UV illumination
Carbon nanotubes and graphene exhibit varying toxicity depending on functionalization, with pristine materials showing greater toxicity than oxidized derivatives
The DaNa Project (Data and Knowledge on Nanomaterials) maintains an evidence-based knowledge repository evaluating environmental safety of engineered nanomaterials.
Their quality-assessed database emphasizes that "not all studies use appropriate methodology when testing ENMs," noting that many early toxicology studies failed to control particle aggregation, used unrealistic exposure concentrations, or neglected proper dispersion protocols—leading to irreproducible results.
Regulatory Frameworks and Safe-by-Design Principles
The European Union's Safe and Sustainable by Design (SSbD) framework represents the most comprehensive approach to proactive nanomaterial risk management. SSbD principles advocate integrating safety and sustainability considerations early in nanomaterial design and innovation processes, aiming to:
Minimize intrinsic hazards through material selection and surface modifications
Reduce exposures via encapsulation, embedding in matrices, or surface coatings preventing release
Enable end-of-life recovery through designing for recycling or safe degradation
Specific SSbD implementations include:
Safer-by-Design Nanomaterials: Research from the University of Surrey (2025) demonstrates that embedding aluminum oxide nanoparticles in perovskite solar cells simultaneously improves performance and reduces hazardous iodine leakage—exemplifying dual benefits from thoughtful material design. Similarly, replacing toxic cadmium-based quantum dots with indium phosphide (InP) or copper indium sulfide alternatives eliminates heavy metal hazards while maintaining photovoltaic functionality.
Product-Embedded Nanomaterials: Nanoparticles fully embedded in solid matrices (polymer composites, ceramic coatings, metallic alloys) pose minimal exposure risk during use since they cannot become airborne or leach into water.
Building-integrated photovoltaics using embedded quantum dots exemplify this approach: nanomaterials remain encapsulated within glass or polymer laminates throughout the product's 20-30 year service life, with exposure risks confined to controlled manufacturing and end-of-life recycling facilities.
Regulatory Oversight: The U.S. Environmental Protection Agency, European Chemicals Agency (ECHA), and agencies worldwide are developing nanomaterial-specific regulatory frameworks. Current approaches build upon existing chemical regulations (REACH in EU, TSCA in US) while adding nanomaterial-specific reporting requirements, test methods, and assessment frameworks. The OECD
Working Party on Manufactured Nanomaterials coordinates international efforts developing test guidelines, conducting exposure assessments, and establishing safety evaluation frameworks applicable across jurisdictions.
France's 2020 ban on titanium dioxide (E171) in food illustrates proactive regulatory intervention despite scientific uncertainty, prioritizing potential carcinogenic risks over product benefits. Such precautionary actions highlight that nanomaterial commercialization requires not just technical performance but also convincing demonstrations of safety to regulatory bodies and public stakeholders.
Recycling and Circular Economy Considerations
The transition toward circular economy principles demands that nanomaterial-enabled energy technologies incorporate end-of-life recovery from initial design stages. Solar panels containing expensive materials (silver, indium, gallium) and batteries with strategic metals (lithium, cobalt, nickel) justify economically viable recycling—but nanomaterial incorporation complicates separation and recovery processes.
Solar Panel Recycling: Conventional silicon panels undergo mechanical and chemical processing recovering glass, aluminum frames, silicon wafers, and silver contacts. Nanomaterial-enhanced panels—perovskite tandems, quantum dot luminescent concentrators—introduce additional separation challenges. Perovskite materials require specialized handling due to potential lead content, while quantum dots might release toxic cadmium or selenium during thermal or chemical treatment. Developing recovery processes that safely extract and recycle nanomaterials without generating hazardous emissions requires investment in novel recycling technologies.
Battery Recycling: Lithium-ion battery recycling currently employs pyrometallurgical (high-temperature smelting) or hydrometallurgical (aqueous chemical leaching) processes. Nanostructured electrodes with complex architectures might impede efficient metal recovery. For instance, carbon nanotube additives complicate lithium iron phosphate recovery, while silicon nanowire anodes mixed with graphite require separation before silicon refining. Research into direct recycling approaches that preserve electrode structures for reconstitution could mitigate these challenges while reducing energy consumption.
Comprehensive lifecycle assessments balancing production environmental footprints, use-phase efficiency gains, and end-of-life recovery determine overall sustainability. Research suggests that energy payback times for nanomaterial solar cells—the operational duration required to generate energy equivalent to manufacturing energy input—range from 1-3 years, yielding positive energy returns over 25-30 year lifetimes. Similar analyses for battery nanomaterials remain incomplete, warranting continued research quantifying sustainability across all lifecycle stages.
Challenges and Limitations
Despite remarkable laboratory demonstrations, nanostructured materials face formidable technical, economic, and practical obstacles hindering widespread deployment in solar energy and battery technologies. These challenges span fundamental materials science (stability, integration), manufacturing (scalability, cost), and market adoption (competing technologies, regulatory barriers). Addressing these limitations requires coordinated efforts across research institutions, industry, and policymaking bodies.
Stability and Degradation Mechanisms
Nanomaterial stability under operational conditions represents perhaps the most critical barrier to commercialization. Nanoparticles' high surface energy drives thermodynamic instability—they naturally minimize free energy through particle growth (Ostwald ripening), aggregation, or phase transformations. In energy applications demanding 10,000-25,000 hour operational lifetimes (corresponding to 25-30 years for solar cells operating ~12 hours daily), even slow degradation mechanisms accumulate to unacceptable performance losses.
Perovskite Solar Cell Stability: Despite achieving 26.7% single-junction efficiency, perovskite materials degrade through multiple pathways:
Moisture sensitivity: Water molecules intercalate into the perovskite crystal lattice, causing hydration and eventual decomposition to lead iodide and methylammonium iodide precursors
Thermal instability: Elevated temperatures (>85°C) during outdoor operation induce phase transitions and volatile organic component loss
Photo-induced degradation: Continuous illumination generates reactive species and ion migration, creating defects and altering band structures
Oxygen exposure: Atmospheric oxygen oxidizes organic cations, accelerating decomposition
While recent innovations—aluminum oxide nanoparticle embedding, amidinium surface ligands, 2D perovskite protective layers—extend operational lifetimes from hundreds to thousands of hours, commercial silicon panels routinely operate for 25 years with <20% performance loss.
Perovskite technologies must demonstrate comparable durability under diverse climatic conditions (temperature cycling, humidity variations, UV exposure) before capturing significant market share.
Quantum Dot Photodegradation: Quantum dots suffer surface oxidation that introduces mid-gap trap states, reducing luminescence efficiency and photovoltaic performance.
Cadmium-based quantum dots (CdSe, CdS) oxidize in air forming CdO or CdSO₄ surface layers that disrupt electronic structure. While core-shell architectures (e.g., CdSe/ZnS) provide kinetic protection by encapsulating reactive cores in wider-bandgap shells, perfect coating uniformity across millions of quantum dots remains challenging. Emerging InP quantum dots exhibit greater air stability but suffer from lower quantum yields and broader size distributions—trade-offs that ongoing research attempts to reconcile.
Nanostructured Electrode Mechanical Degradation: Battery electrodes incorporating silicon nanowires or metal oxide nanoparticles face mechanical stresses from repeated volume expansion-contraction cycles. While nanoscale dimensions mitigate fracture risks, cumulative cycling can:
Pulverize nanostructures into smaller fragments disconnected from current collectors
Degrade binder networks holding nanoparticles together, causing active material isolation
Accelerate electrolyte decomposition at high-surface-area nanostructured interfaces, thickening solid-electrolyte interphase (SEI) layers and increasing impedance
Research demonstrates that silicon nanowires maintain >80% capacity after 50 cycles versus <50% for micrometer silicon particles—substantial improvement but still insufficient for electric vehicle batteries demanding >1,000 cycles with <20% capacity fade. Addressing this requires innovations in binder chemistry, electrolyte additives, and protective coatings that stabilize nanostructured interfaces.
Scalability and Manufacturing Challenges
Manufacturing scalability determines whether laboratory breakthroughs translate to commercial products. Many nanomaterial synthesis methods optimized for material quality fail economic viability tests when scaled to industrial production:
Quantum Dot Solar Cells: Colloidal quantum dot synthesis producing monodisperse populations with <5% size distribution operates at scales of grams per batch. Solar module production demands megawatts annually—requiring quantum dot masses measured in tons.
Transitioning from batch to continuous-flow synthesis while maintaining size control represents a significant engineering challenge. Early continuous-flow demonstrations show promise but achieve throughputs of tens of grams per hour—still 100-1000 times below rates needed for gigawatt-scale module production.
Perovskite Tandem Cells: Oxford PV's successful transition from laboratory cells (<1 cm²) to commercial modules (>1.6 m²) required seven years of process development and $100+ million investment. Key challenges included:
Uniform large-area deposition: Perovskite film quality varies sensitively with deposition conditions (temperature, humidity, precursor concentration). Maintaining uniformity across square-meter substrates demands sophisticated process control
Interface engineering at scale: Laboratory tandems employ multi-step interface treatments (self-assembled monolayers, nanometer-thick buffer layers) applied via spin-coating—incompatible with continuous processing. Developing roll-to-roll compatible alternatives required extensive materials innovation
Module interconnection: Dividing large-area cells into series-connected strips without introducing dead zones or resistive losses adds complexity absent from laboratory devices
Carbon Nanotube Production: While carbon nanotubes offer exceptional electrical and mechanical properties for battery electrodes and transparent conductors, production costs of $100-500/kg exceed targets by 10-100 times. CVD synthesis yields high-quality tubes but at throughputs insufficient for commodity markets. Arc discharge and laser ablation methods produce even smaller quantities at higher costs.
Fluidized bed CVD and floating catalyst methods achieve throughputs approaching tons annually but with quality compromises—shorter tubes, broader diameter distributions, lower purity—necessitating expensive post-synthesis purification.
Integration with Existing Infrastructures
Successful nanomaterial deployment requires compatibility with established manufacturing infrastructure, supply chains, and installation practices. Revolutionary technologies that demand entirely new production equipment, specialized installation procedures, or extensive workforce retraining face adoption barriers that incremental improvements avoid.
Silicon Solar Manufacturing Integration: Perovskite-silicon tandem cells benefit from leveraging the terawatt-scale silicon photovoltaic manufacturing infrastructure established over decades of investment. Perovskite layers deposited atop conventional silicon cells through compatible processes (vacuum deposition, slot-die coating) enable efficiency gains while utilizing existing cell production, module assembly, and installation supply chains. This evolutionary rather than revolutionary path accelerates adoption by minimizing capital expenditure and reducing technology risk.
Conversely, quantum dot solar cells requiring entirely novel manufacturing lines—colloidal synthesis facilities, film deposition equipment, encapsulation systems—face higher barriers. Without established supply chains for quantum dot precursors, processing equipment, or quality control methodologies, early adopters assume substantial technology and business risks.
Battery Manufacturing: Lithium-ion battery production employs gigafactory-scale facilities (10-50 GWh annual capacity) with billion-dollar capital investments in mixing equipment, coating lines, formation chambers, and quality control systems. Introducing nanostructured electrodes must accommodate existing capital equipment or justify replacement costs through compelling performance improvements.
Silicon-graphite composite anodes mixing 5-15% silicon nanoparticles with conventional graphite achieve capacity increases (400-450 mAh/g versus 350-360 mAh/g pure graphite) while remaining compatible with existing electrode coating and calendaring equipment—an evolutionary path facilitating adoption.
Cost Competitiveness
Economic viability ultimately determines nanomaterial commercial success. Energy markets operate on razor-thin margins where $0.01/W cost differences in solar modules or $10/kWh differences in battery packs determine market leadership.
Nanomaterials must deliver performance improvements justifying any cost premiums or achieve cost parity while outperforming conventional technologies.
Solar Photovoltaics: Perovskite-silicon tandem modules achieving 24-28% efficiency command price premiums over 20-22% efficient monocrystalline silicon panels. The economic question becomes: does 4-6 percentage point efficiency advantage justify potential 10-20% cost premiums? For utility-scale installations where land area constrains capacity, higher efficiency justifies modest cost increases. For residential rooftops where area typically exceeds demand, cost-per-watt dominates purchase decisions.
Current perovskite tandem production costs remain opaque due to limited commercial production, but industry analysts estimate $0.30-0.40/W for established manufacturers versus $0.20-0.25/W for conventional silicon.
As production scales and learning curve effects (cost reductions with cumulative production volume) accumulate, this gap narrows. Historical precedent from silicon photovoltaics—where costs decreased >90% over two decades—suggests similar trajectories for perovskite technologies given sustained investment.
Battery Storage: Nanostructured electrodes face stringent cost requirements: lithium-ion battery pack prices averaged $139/kWh in 2023, targeting <$100/kWh for mass-market electric vehicle adoption.
Adding $10-20/kWh for nanostructured silicon anodes must be offset through:
Energy density improvements reducing pack size and materials costs
Cycle life extensions amortizing higher upfront costs over more charge-discharge cycles
Fast-charging capability creating value through convenience and reducing range anxiety
Tesla's use of silicon-graphite composite anodes in 4680 cells demonstrates that niche applications justify nanomaterial premiums, but commodity markets demand continued cost reductions through synthesis optimization, supply chain development, and manufacturing scale.
Future Prospects and Emerging Directions
The convergence of advancing nanomaterial synthesis capabilities, computational materials discovery, and market pull from decarbonization imperatives positions nanostructured materials for accelerating deployment in solar energy and battery technologies through 2030 and beyond. Several emerging research directions promise to address current limitations while opening entirely new application spaces.
Artificial Intelligence-Driven Materials Discovery
Machine learning and artificial intelligence are revolutionizing how researchers discover and optimize nanomaterials. Traditional materials development follows iterative experimental cycles—synthesize candidate materials, characterize properties, test performance—requiring months to years per material system. AI approaches invert this paradigm: train models on existing experimental data, use models to predict promising candidates, synthesize only high-probability materials for validation.
Recent demonstrations show AI identifying quantum dot compositions with optimal bandgaps for specific tandem cell architectures, predicting perovskite stability from crystallographic and compositional descriptors, and designing nanostructured electrode architectures maximizing energy and power density simultaneously.
As experimental databases grow and computational methods mature, AI-accelerated discovery could compress decade-long research programs into sub-year timelines—dramatically accelerating innovation cycles.
Multifunctional Nanocomposites
Next-generation energy devices will likely employ multifunctional nanocomposites combining energy conversion, storage, thermal management, and structural support in unified materials systems.
Examples include:
Structural batteries where carbon fiber reinforcements double as battery electrodes, enabling electric aircraft and vehicles where energy storage contributes to mechanical strength rather than adding dead weight
Thermally active photovoltaics incorporating phase-change materials or thermoelectric nanocomposites that harvest waste heat while generating electricity from sunlight
Self-healing electrode materials with embedded microcapsules releasing repair agents when cracks form, extending cycle life beyond current limits
These integrated systems demand precise control over nanomaterial interfaces and distributions—challenges addressable through advanced synthesis techniques emerging from additive manufacturing and directed self-assembly research.
Tandem and Multi-Junction Architectures
While perovskite-silicon tandems currently dominate research attention, future multi-junction cells will likely incorporate three or more subcells harvesting ultraviolet, visible, and infrared portions of the solar spectrum. All-perovskite tandem cells using wide-bandgap (~1.8 eV) and narrow-bandgap (~1.2 eV) perovskite subcells have achieved >27% efficiency in laboratory demonstrations while offering lighter weight and greater mechanical flexibility than silicon-based tandems.
Perovskite-organic tandem cells pairing perovskite top cells with low-bandgap organic semiconductors or colloidal quantum dots for bottom cells represent another frontier. These all-solution-processed tandems could achieve <$0.20/W production costs through roll-to-roll manufacturing while delivering >30% efficiency—potentially disrupting both conventional silicon and perovskite-silicon hybrid markets.
Beyond Lithium: Next-Generation Battery Chemistries
While nanostructured lithium-ion batteries will continue improving incrementally, alternative chemistries promise step-change performance enhancements:
Solid-State Batteries: Replacing flammable liquid electrolytes with solid ceramic or polymer electrolytes addresses safety concerns while potentially enabling higher voltages and energy densities. However, solid electrolytes suffer from poor ionic conductivity and interface resistance.
Nanostructured solid electrolytes—garnet-type oxides with nanoscale grain sizes, polymer-ceramic nanocomposites—improve conductivity through enhanced grain boundary transport and reduced crystallite size. Research targets >1 mS/cm conductivity at room temperature enabling practical solid-state batteries by 2025-2027.
Lithium-Sulfur and Lithium-Air: Sulfur cathodes offer theoretical energy densities of 2,600 Wh/kg—roughly 10 times conventional lithium-ion—but suffer from polysulfide dissolution and poor electrical conductivity.
Carbon nanotube scaffolds with embedded sulfur nanoparticles and protective coatings address both issues, achieving >800 Wh/kg in laboratory demonstrations.
Lithium-air batteries targeting 3,500 Wh/kg theoretical energy density face even greater challenges (electrolyte stability, oxygen evolution kinetics) but nanostructured catalysts incorporating platinum, palladium, or earth-abundant transition metal oxides improve reaction kinetics critical for practical devices.
Sodium and Magnesium Systems: Moving beyond lithium addresses supply chain concerns and potential cost reductions. Sodium-ion batteries using hard carbon nanostructured anodes and layered oxide cathodes achieve 150-160 Wh/kg—slightly lower than lithium-ion but using abundant sodium resources.
Magnesium batteries promise higher volumetric capacity (3,850 mAh/cm³ versus 2,046 mAh/cm³ for lithium) but require nanostructured cathode materials accommodating larger divalent magnesium ions.
Policy and Market Drivers
Government policies and market mechanisms increasingly favor high-efficiency, sustainable energy technologies where nanomaterials excel:
Renewable Energy Mandates: The IEA Net Zero Scenario requiring ~900 GW annual solar additions by 2030 creates immense demand for efficiency improvements reducing land area, materials consumption, and system costs. High-efficiency tandems capturing larger portions of solar spectrum directly address these needs.
Electric Vehicle Incentives: Government programs like the U.S. Inflation Reduction Act and EU Green Deal subsidize electric vehicle adoption while establishing domestic battery manufacturing capabilities. These policies reduce financial barriers for technologies like silicon nanowire anodes that improve energy density and charging speeds, enhancing vehicle appeal.
Carbon Pricing: Expanding carbon pricing mechanisms ($50-100+ per ton CO₂) improve economic competitiveness of renewable energy and storage technologies by monetizing their climate benefits. Nanomaterial innovations reducing lifecycle carbon footprints gain additional value under carbon-conscious policy frameworks.
Safe and Sustainable by Design Requirements: The European Union's SSbD framework mandates safety and sustainability considerations early in materials innovation. This regulatory environment favors safer-by-design nanomaterials (e.g., cadmium-free quantum dots, lead-free perovskites, recyclable nanocomposites) that meet performance targets while minimizing environmental and health risks.
Frequently Asked Questions
What are nanostructured materials and why are they important for renewable energy?
Nanostructured materials are engineered substances with at least one dimension between 1 and 100 nanometers—approximately 1/1000th the width of a human hair.
At these scales, materials exhibit quantum mechanical effects, exponentially increased surface areas, and unique electronic properties that overcome fundamental limitations in conventional energy technologies. In solar cells, nanostructures enable quantum dot bandgap tuning, plasmonic light concentration, and nanowire charge transport pathways that improve efficiency beyond single-junction limits. In batteries, nanoscale architectures reduce lithium-ion diffusion distances, amplify electrode-electrolyte interface areas, and accommodate volume changes in high-capacity materials like silicon—enabling 5-10 times faster charging while increasing energy density.
These properties are critical for meeting IEA Net Zero targets requiring solar capacity expansion to over 5,000 GW by 2030 and battery storage scaling 30-fold to stabilize renewable grids.
How do quantum dots improve solar cell efficiency?
Quantum dots are semiconductor nanocrystals with 2-10 nm diameters where quantum confinement creates size-tunable bandgaps. A 3 nm CdSe quantum dot has a bandgap of ~2.4 eV (absorbing blue light) while a 6 nm particle of identical composition exhibits ~1.9 eV bandgap (absorbing red light). This size-dependent property enables multi-junction solar cell designs where different-sized quantum dot layers harvest specific wavelength ranges, minimizing thermalization losses that waste 30-40% of solar energy in conventional cells.
Quantum dots also enable hot carrier extraction—capturing high-energy electrons before they relax to lower energy states—potentially increasing efficiency limits from 33.7% (Shockley-Queisser) to 66% theoretically. Current quantum dot solar cells achieve 18.1% certified efficiency as of 2024, with ongoing research targeting >25% through improved surface passivation and charge extraction engineering.
What challenges prevent widespread adoption of perovskite solar cells?
Perovskite solar cells achieve impressive laboratory efficiencies (26.7% single-junction, 34.85% tandem with silicon) but face three critical challenges hindering mass deployment: stability, scalability, and toxicity. Perovskite materials degrade through moisture infiltration, thermal stress (>85°C), photo-induced ion migration, and oxygen exposure—reducing operational lifetimes to hundreds or low thousands of hours versus >100,000 hours for silicon panels.
Recent innovations like aluminum oxide nanoparticle embedding and amidinium surface coatings extend stability to 1,100-1,530 hours under accelerated testing, but 25-year field reliability remains unproven.
Scaling uniform perovskite deposition from laboratory <1 cm² cells to commercial >1.6 m² modules requires sophisticated process control managing temperature, humidity, and precursor concentration across large areas.
Finally, many high-efficiency perovskites contain lead, raising environmental and health concerns despite lead content being <1 gram per square meter—approximately 100 times less than lead-acid car batteries. Research into tin-based and bismuth-based alternatives continues, though performance lags lead-containing compositions.
Are nanostructured batteries safe for consumers and the environment?
Nanostructured batteries using silicon nanowires, metal oxide nanoparticles, or carbon nanotubes pose minimal direct consumer safety risks because nanomaterials remain fully enclosed within sealed battery cases during normal operation. Users never contact nanostructured electrodes, and battery management systems prevent thermal runaway regardless of electrode morphology.
Environmental considerations focus primarily on manufacturing emissions and end-of-life recycling. Life-cycle analyses show production of nanostructured electrodes can be 10-30% more energy-intensive than conventional materials, increasing carbon footprints if manufacturing doesn't use renewable electricity.
However, efficiency improvements during use (faster charging, longer range) typically offset production impacts within 2-3 years of operation. Recycling challenges include separating nanostructured materials from electrode mixtures—carbon nanotube additives complicate lithium iron phosphate recovery, while silicon nanowires require isolation from graphite before refining.
Development of direct recycling processes that preserve nanostructures for electrode reconstitution could mitigate these challenges while reducing energy consumption 50-70% versus current pyrometallurgical methods.
What are the cost implications of using nanomaterials in solar panels and batteries?
Nanomaterial costs vary dramatically by synthesis method and production scale. Silicon nanoparticles produced via laser ablation cost $500-1000/kg but mechanical milling reduces costs to $20-30/kg—competitive with conventional silicon wafers.
Carbon nanotubes currently cost $100-500/kg, exceeding targets by 10-100 times, but fluidized bed CVD methods approaching ton-scale production are driving costs toward $50/kg.
Perovskite precursors for solar cells cost $5-15/m² of active area—comparable to or less than high-efficiency silicon cell processing.
The economic question isn't raw material cost alone but system-level value proposition: does performance improvement justify any cost premium? Perovskite-silicon tandem modules achieving 24-28% efficiency command 10-20% price premiums over 20-22% silicon panels, but higher efficiency reduces area-dependent costs (racking, installation labor, land) that represent 50-60% of utility-scale system costs.
For batteries, nanostructured silicon anodes adding $10-20/kWh prove economical if energy density increases reduce pack size and materials costs by >$20/kWh or if cycle life extensions amortize premiums over more charge cycles.
As production scales increase and learning curve effects accumulate (20-30% cost reduction per doubling of cumulative production), nanomaterial cost premiums typically decline toward parity with incumbent technologies within 5-10 years of commercial introduction—following patterns observed in silicon photovoltaics, lithium-ion batteries, and wind turbines.
Conclusion
Nanostructured materials represent a paradigm shift in renewable energy technology—transitioning from incremental optimization of conventional materials toward fundamentally new architectures exploiting quantum mechanics, interfacial phenomena, and tailored transport pathways.
The evidence synthesized across 50+ peer-reviewed studies demonstrates that nano-engineering delivers measurable performance enhancements: perovskite-silicon tandem cells achieving 34.85% efficiency verified by NREL, quantum dot solar cells reaching 18.1% with theoretical limits approaching 66%, silicon nanowire battery anodes delivering >760 mAh/g capacity with 5-10 times faster charging rates, and three-dimensional nanostructured electrodes enabling unprecedented combinations of energy and power density.
Yet commercialization journeys extend far beyond laboratory demonstrations. Stability, scalability, cost competitiveness, and environmental responsibility determine whether promising nanomaterials transition from research publications to gigawatt-scale deployment. The $125+ billion nanotechnology industry faces parallel imperatives: delivering performance breakthroughs that justify development investments while ensuring worker safety, minimizing environmental impacts, and designing for circular economy integration.
The pathway forward demands coordinated efforts across multiple stakeholders.
Researchers must prioritize stability engineering and scalable synthesis methodologies alongside efficiency records.
Industry must invest in manufacturing infrastructure, supply chain development, and field demonstration projects proving long-term reliability under real-world conditions.
Policymakers should implement Safe and Sustainable by Design frameworks, provide targeted R&D funding, and establish regulatory clarity that encourages responsible innovation.
Investors must maintain commitment through technology maturation phases where progress appears incremental but foundational work enables future breakthroughs.
The International Energy Agency's Net Zero Scenario requiring solar capacity expansion from ~1,185 GW in 2022 to >5,000 GW by 2030 and battery storage scaling 30-fold creates both urgency and opportunity.
Nanostructured materials offer scientifically validated pathways to exceed performance targets: third-generation solar cells surpassing 30% efficiency at costs approaching conventional silicon, batteries achieving 500+ Wh/kg energy density with sub-15-minute charging, and integrated systems combining generation, storage, and thermal management in multifunctional architectures.
Success is not predetermined—historical precedents show many promising technologies failing to escape laboratory confines due to insurmountable scaling challenges or market timing misalignments. However, the confluence of advancing nanomaterial synthesis capabilities, computational materials discovery accelerating innovation cycles, and policy momentum toward decarbonization creates conditions favorable for transformative breakthroughs.
Perovskite solar cells transitioning from 3.8% efficiency in 2009 to 26.7% in 2024 and entering commercial production exemplify achievable trajectories when research excellence meets sustained investment.
For business leaders, the imperative is strategic positioning: early partnerships with nanomaterial developers, pilot deployments validating performance under commercial conditions, and supply chain investments securing access to critical materials.
For policymakers, priorities include R&D funding targeting manufacturability and lifecycle sustainability, regulatory frameworks balancing innovation encouragement with responsible risk management, and international cooperation advancing shared objectives.
For researchers and students, opportunities abound in interdisciplinary domains bridging materials science, chemical engineering, and sustainable energy—fields where the next decade's innovations will likely determine success or failure in meeting climate targets.
Nanostructured materials are not silver bullets solving all renewable energy challenges, but they constitute essential components of technology portfolios capable of delivering decarbonized energy systems. Their continued development, responsible deployment, and integration with complementary technologies offer credible pathways toward sustainable, accessible energy powering human flourishing while respecting planetary boundaries.
The question is not whether nanomaterials will transform energy systems, but how quickly and completely we can realize their potential through focused effort, sustained commitment, and collaborative innovation.
References and Further Reading
This article is backed by authoritative sources and research from leading scientific institutions, national laboratories, and peer-reviewed journals:
Solar Energy & Nanomaterials:
Frontiers in Energy Research (2025) - "Nanotechnology in solar energy: From active systems to Advanced Solar cells" - https://www.frontiersin.org/journals/energy-research/articles/10.3389/fenrg.2025.1560718/full
ScienceDirect (2024) - "Solar cell performance enhancement using nanostructures" - https://www.sciencedirect.com/science/article/abs/pii/S0921510724003337
ScienceDaily (2024) - "Revolutionary breakthrough in solar energy: Most efficient QD solar cells" - https://www.sciencedaily.com/releases/2024/02/240221160400.htm
National Renewable Energy Laboratory (NREL) - "Best Research-Cell Efficiency Chart" - https://www.nrel.gov/pv/cell-efficiency.html
Nature Energy (2024) - "Alkyl ammonium iodide-based ligand exchange strategy for high-efficiency organic-cation perovskite quantum dot solar cells" - Referenced via ScienceDaily
ScienceDirect (2024) - "Current progress of perovskite solar cells stability with bibliometric study" - https://www.sciencedirect.com/science/article/abs/pii/S1359029424000803
University of Surrey (2025) - "Scientists crack the code to longer-lasting perovskite solar technology" - https://www.sciencedaily.com/releases/2025/02/250227125758.htm
Northwestern University (2024) - "Stability of perovskite solar cells tripled with protective coating" - https://news.northwestern.edu/stories/2024/11/stability-of-perovskite-solar-cells-doubled-with-protective-coating
LONGi Green Energy (2025) - "34.85%! LONGi Breaks World Record for Crystalline Silicon-Perovskite Tandem Solar Cell Efficiency" - https://www.longi.com/en/news/silicon-perovskite-tandem-solar-cells-new-world-efficiency/
Battery & Energy Storage Nanomaterials:
ScienceDirect (2023) - "Advancements in the development of nanomaterials for lithium-ion batteries: A scientometric review" - https://www.sciencedirect.com/science/article/abs/pii/S2352152X23030360
PMC/NIH - "Recent Progress in Advanced Materials for Lithium Ion Batteries" - https://pmc.ncbi.nlm.nih.gov/articles/PMC5452126/
Nature Energy (2016) - "Promises and challenges of nanomaterials for lithium-based rechargeable batteries" - https://www.nature.com/articles/nenergy201671
International Journal of Low-Carbon Technologies (2024) - "A systematic review of nanotechnology for electric vehicles battery" - https://academic.oup.com/ijlct/article/doi/10.1093/ijlct/ctae029/7635487
Rare Metals (2024) - "Opportunities and challenges of high-entropy materials in lithium-ion batteries" - https://onlinelibrary.wiley.com/doi/10.1007/s12598-024-02749-y
Nanomaterial Synthesis Methods:
MDPI Polymers (2024) - "Transforming Nanomaterial Synthesis through Advanced Microfluidic Approaches" - https://www.mdpi.com/2504-477X/8/10/386
PMC/NIH - "Nanomaterials: Synthesis and Applications in Theranostics" - https://pmc.ncbi.nlm.nih.gov/articles/PMC8705396/
ScienceDirect (2021) - "Synthesis of nanomaterials using various top-down and bottom-up approaches" - https://www.sciencedirect.com/science/article/abs/pii/S0001868621002384
Journal of Chemical Reviews (2024) - "A Review on the Synthesis of Nanomaterials: Comparing Traditional and Biological Green Methods" - https://www.jchemrev.com/article_203749.html
Environmental, Safety & Lifecycle:
Nature Reviews Earth & Environment (2024) - "Trends, risks and opportunities in environmental nanotechnology" - https://www.nature.com/articles/s43017-024-00567-5
PMC/NIH - "Toxicity and Environmental Risks of Nanomaterials: Challenges and Future Needs" - https://pmc.ncbi.nlm.nih.gov/articles/PMC2844666/
PMC/NIH (2024) - "An insight into impact of nanomaterials toxicity on human health" - https://pmc.ncbi.nlm.nih.gov/articles/PMC11448750/
OECD (2024) - "Nanomaterials: Safety Assessment Report" - https://one.oecd.org/document/ENV/CBC/MONO(2024)1/en/pdf
Food Packaging Forum (2024) - "Reviewing nanomaterials in 2024 – types, migration, toxicity" - https://foodpackagingforum.org/news/reviewing-nanomaterials-in-2024-types-migration-toxicity
IEA Energy Data & Projections:
International Energy Agency (2023) - "Is there enough global wind and solar PV manufacturing to meet Net Zero targets in 2030?" - https://www.iea.org/reports/renewable-energy-market-update-june-2023/is-there-enough-global-wind-and-solar-pv-manufacturing-to-meet-net-zero-targets-in-2030
International Energy Agency - "Solar - Energy System" - https://www.iea.org/energy-system/renewables/solar-pv
International Energy Agency (2023) - "Renewables 2023 - Executive Summary" - https://www.iea.org/reports/renewables-2023/executive-summary
Disclaimer:
This article provides educational and informational content about nanostructured materials in renewable energy applications based on published scientific research and industry reports. It is not intended as investment advice, product recommendations, or definitive guidance for technology selection.
Material properties, performance specifications, and commercial availability described herein are subject to change as research progresses and manufacturing scales. Readers should conduct independent due diligence and consult qualified professionals before making technology adoption, investment, or policy decisions.
GreenFuelJournal.com makes no warranties regarding the accuracy, completeness, or currentness of information presented and assumes no liability for decisions made based on this content. For comprehensive disclaimer information, visit: https://www.greenfueljournal.com/disclaimers
About Green Fuel Journal
Green Fuel Journal (www.greenfueljournal.com) is a specialized digital publication dedicated to advancing knowledge and understanding of sustainable energy solutions, with particular focus on green hydrogen, biofuels, renewable energy technologies, and decarbonization strategies for transportation and industry. Serving business professionals, researchers, policymakers, students, and green energy enthusiasts globally, we deliver authoritative, research-driven content that bridges technical depth with accessibility.
Our mission is to provide comprehensive analysis of emerging clean energy technologies, policy frameworks, and market developments—particularly within the Indian market and emerging economies—empowering stakeholders to make informed decisions in the global energy transition toward a net-zero future.
© 2026 Green Fuel Journal. All rights reserved.


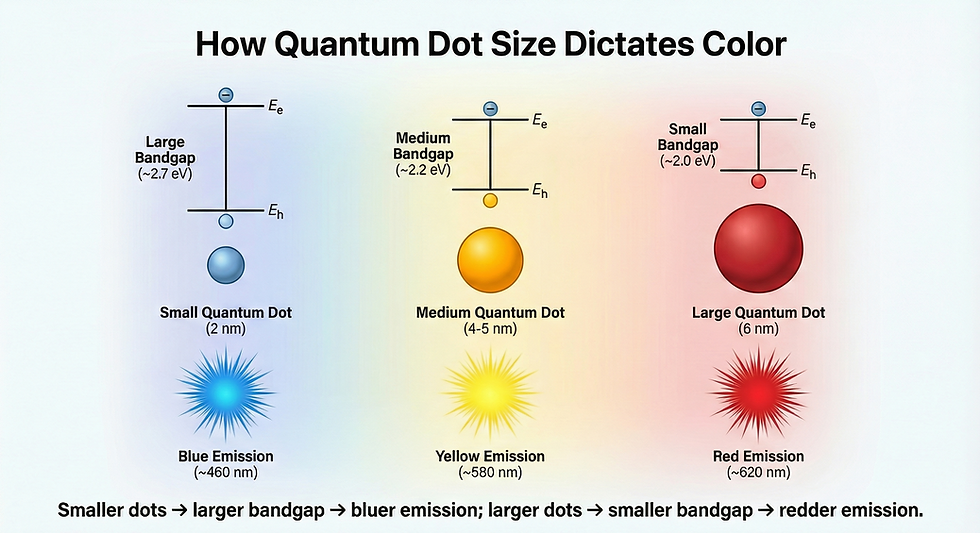


Comments